∞ C.S.T.I .∞
開發高性能、安全可靠的細胞培養基,以支持細胞療法和生物製藥
開發傑出的高端無血清琣養的技術
細胞科學技術研究所使用先進的動物細胞培養技術開發了獨特的高性能無血清培養基。 這些技術也獲得了外部研究機構的高度評價,使我們能夠參與眾多聯合研發和公共聯合研究項目。 我們的目標是通過支持細胞培養技術為社會做出貢獻,該技術在近年來不斷發展的再生醫學/細胞療法和藥物生產研究領域以及基礎研究中發揮著越來越重要的作用,並通過 提供高性能的無血清培養基。

CEO致辭
自從首次成功報導細胞培養成功以來,已有近100年的歷史了。在此期間,由於眾多研究人員的努力,細胞培養技術取得了重大進展。當前,在細胞培養技術上尋求進一步的進步,該技術作為支持再生醫學/細胞療法以及作為醫療保健的未來的生物藥物生產的基礎技術。細胞培養基對於細胞培養是必不可少的,可以毫不誇張地說,它們的性能決定了細胞培養的成敗。自2001年成立以來,我們是一家專門從事細胞培養基的公司,我們不僅為基礎研究提供了安全,高性能的細胞培養基,還為進行尖端細胞培養的領域(例如再生醫學研究)提供了安全,高性能的細胞培養基/細胞療法和生物製藥製造。我們相信,作為NIPRO Group的一員,我們公司有責任支持醫療保健的未來,將產品開發向前一步,以安全為重中之重。


產品介紹


1.用於細胞的無血清培養基以及再生醫學/細胞療法的研究
我們改進和新開發了無血清培養基,用於免疫細胞治療和用於細胞治療的成纖維細胞。
2.用於乾細胞研究的無血清培養基
我們開發無血清培養基,用於培養iPS細胞(誘導性多能幹細胞)和MSC(間充質乾細胞),它們有可能成為再生醫學/細胞療法研究的領先者。
3.用於生產抗體,疫苗和重組蛋白的無血清培養基
利用細胞培養進行生產正成為生產疫苗和醫療用蛋白質的主要技術。 我們開發無血清培養基,旨在將其用於生產此類藥物。
4.化學試劑及其他產品
我們製造和銷售各種類型的細胞培養基和細胞培養相關試劑。
細胞科學研究所は細胞培養液で高度先進醫療の研究をサポートします
●開發用於免疫細胞療法研究的SFM
●開發用於再生醫學研究的SFM
●開發用於生物醫學生產的SFM
●定制/改良培養基的生產
●動物細胞培養的技術支持
使用培養袋系統進行人LAK細胞培養
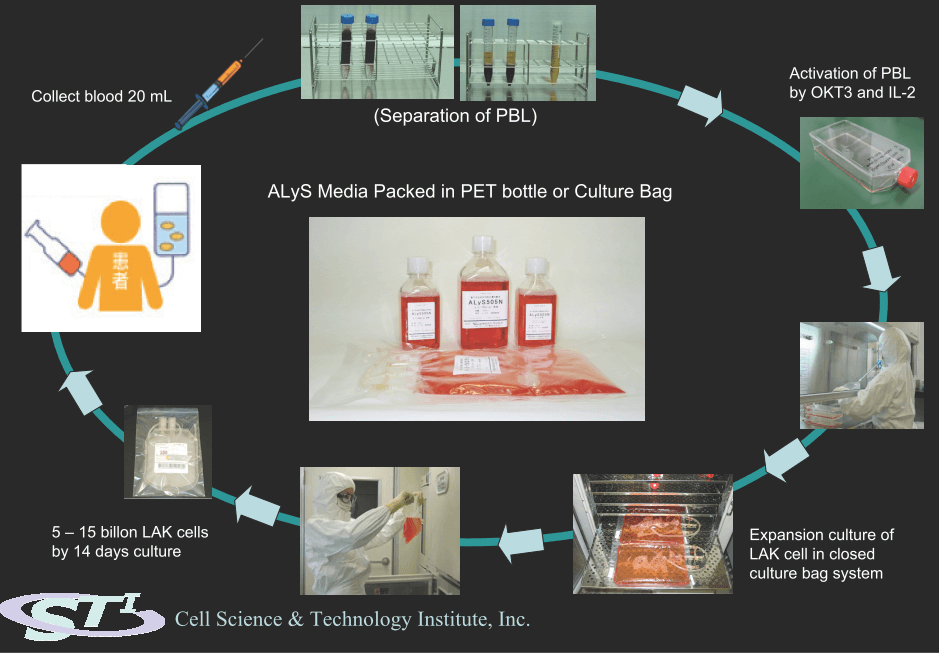
SFM用於癌症免疫細胞治療研究ALyS 505N
1.首選人類LAK,NK和CTL培養
2.所有蛋白質成分均為重組型或醫學級類型
3.提供IL-2預添加培養基
4.基於GMP的生產
5.培養袋預裝
用於淋巴細胞培養的無動物衍生因子培養基ALyS 705
1.優於動物淋巴細胞培養
2.無動物衍生因子
3.基於GMP的生產
ALyS505N對T細胞衝擊波的增殖
普通血清培養基中LAK細胞生長的比較研究
(改良的RPMI-1640)和帶有癌症患者PBL的ALyS505N培養基
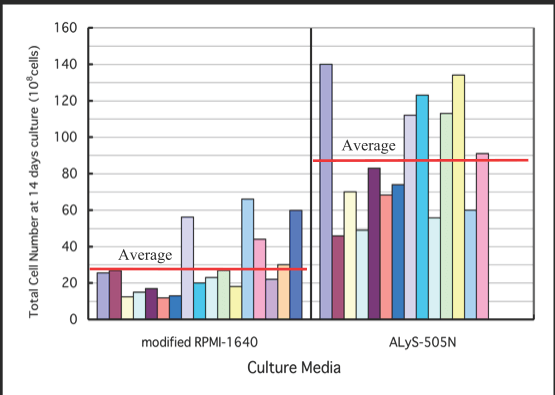
改良的RPMI-1640:10%自體血清(AS)持續6天,然後2%AS持續8天。 ALyS-505N:4天5%AS,然後無血清10天。
ALyS505N用戶在日本,韓國
*主要客戶/位置
瀨田診所集團(瀬田クリニック)/東京,札幌,
INNOMEDYSIS(イノメディシス)/韓國Seouru
生物療法研究所 日本(日本バイオセラピー研究所)/東京都
仙台微生物研究所SENBIKEN Clinic /仙台
東京大學病院/東京大學
東京女子醫科大學附屬醫院/東京
千葉癌中心(千葉県立ガンセンター)/千葉
Immuno Teck Co.,Ltd(株式會社イムノテック)/大阪
統合醫療村/東京
昭壽醫學研究所(福祉村病院長壽醫學研究所)/豐橋
符合GMP的生產設施


細胞處理研發中心







